And why are we so pedantic about keeping it out of our hair and face bars?
When you get down to it, soap is a mixture of oils or fats with a strong alkali that is really good at cleaning! The special composition attracts dirt, oil and bacteria to help wash them away when used with water.
So what is pH?
pH means how acidic or alkaline a substance is. Here is a pH scale:
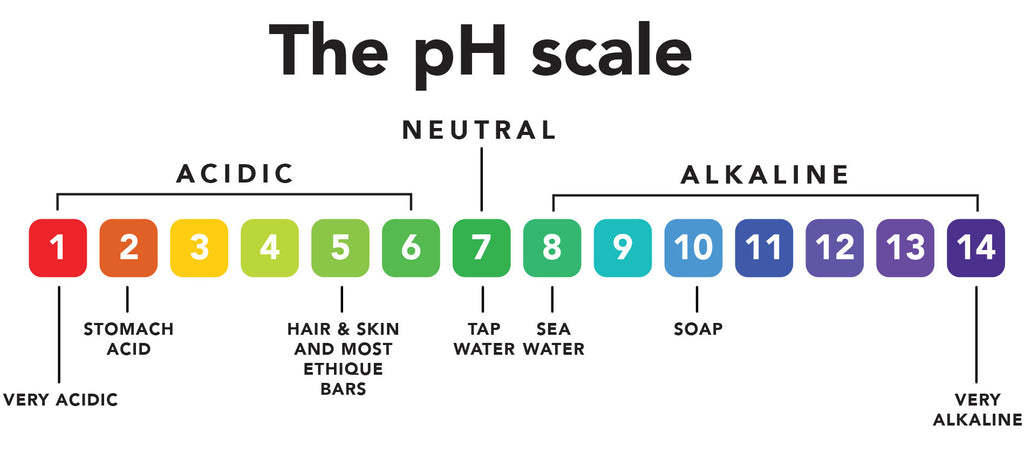
As you can see, 1 is acidic and 14 is very alkaline. Soap sits at 10, meaning it has a high pH.
But why does this actually matter for hair?
It’s a proven fact that products with a high pH damage hair. That’s why we choose to make more expensive, pH-balanced bars for hair and face, instead of more affordable soap-based bars.
If you don’t have sensitive skin or other skin issues, you can usually use soap to wash your body (see more here). Skin has a pH of about 5.5. When you rinse soap off, your acid mantle (secretions from your skin) is washed away, but it replaces itself rapidly – between 20 minutes to an hour after your shower. This acid mantle helps keep pathogens from using your face as an all-you-can-eat buffet, and prevents trans-epidermal water loss (TEWL), which causes dehydrated skin. (For those of you who are unlucky and find soap too drying on your body, a soap free bodywash is a great alternative!)

The bad news is that your hair doesn’t have an acid mantle. The sebum from your scalp doesn’t go very far down your hair, so the hair remains unprotected. Whatever pH product you use on it, is the pH your hair stays for a considerable period. The natural pH of hair is around 4.5-5.5. This acidic pH keeps the cuticles on your hair shaft lying smoothly against the inner cortex. If they don’t lie flat against this inner ‘core’, your hair loses moisture and condition much quicker, looks rough and dull (as light doesn’t bounce off a rough surface) and over time, this causes split ends, loss of hair colour, breakage and severe dryness. Do you ever wonder why they recommend you use a vinegar rinse after using a soap-based shampoo bar? This is exactly why. Vinegar rinses help, but the damage is still done with high pH products.

Okay, so if our bars are soap free and pH balanced, what exactly makes them foam?
Surfactants. Surfactants do exactly the same thing as soap. In fact, soap is a type of surfactant (which stands for surface active agent.) But the beauty of soap free surfactants is that the formulas they are combined in, can have their pH adjusted to the acidic range required by hair and skin, whereas a soap molecule will fall apart and you will be back to just having oil and water.
Some surfactants are natural and some are synthetic, but they are ALL produced in a lab, but as we are not scared of chemicals (remember, you are made up of them), we know that that doesn’t mean anything. Of course, we use only plant derived, gentle surfactants.
How does a surfactant clean?
Magic!
Okay, not magic. But it’s a simple concept. We know oil and water don’t mix as they repel one another. So if you wash your face with water, most of the sebum and makeup will stay where it is. So to enable something to cleanse, you need to ensure it’s able to bind to an oil molecule. But to ensure it’s easy to rinse off, you need to ensure it can also bind to water. How?! By using molecules that can bind to both. The correct term is an amphipathic molecule and it simply means a molecule like the below, which has an oil-loving (hydrophobic) tail and a water-loving (hydrophilic) head.

So when you use your bar on your wet face, the surfactant ‘tail’ binds with sebum and other oily substances (like makeup), whilst the ‘head’ enables spreading and ensures it can be easily rinsed away.
Surfactants are also what makes something foam (and yet some of them don’t foam) and are also emulsifiers to make creams, but that is another topic, for another day. (Interested? Let me know.)
So is soap more natural than soap-free surfactants (or synthetic detergents as they are called)?
Not necessarily. Not only does natural not mean in anyway safe, (arsenic, fluoride and cyanide are all 100% natural and 100% fatal in small doses), but soap can be synthetic and soap-free surfactants can be certified natural. So you do need to take each ingredient on its own merits. We like the Environmental Working Group’s (EWG) certification for surfactants- all of ours are rated at 1, which for those of you unfamiliar, is the highest safety rating EWG gives. Something to bear in mind too, if you are concerned about palm oil, is that by number of bars sold ~99% of the world’s soap contains palm oil in some form. (Look for palm oil, vegetable oil or fat, palm kernelate or sodium palmitate for just a few names), whereas it is possible to source palm free surfactants with some ease. Ours are coconut, sugar and rice bran derived.
Hopefully that has given you an understanding of why we proudly state that our bars are soap free, pH balanced and colour safe. Please send through any questions on anything I haven’t explained clearly.
View our soap free bars here.



